Controlling chronic pain with the Quell companion app

NeuroMetrix is a healthcare technology company revolutionizing pain management. Their FDA-cleared wearable device, Quell, offers drug-free relief to chronic pain sufferers.
Control their therapy and improve their pain management using the app.
Quell Relief users across the globe access the app more than 20,000 times every day.
Reported improvement in chronic pain and overall health in an Iposos study.
Client Outcomes

“I’m very happy with the app. I think it adds a lot of value. Now that we’re on the market, we have people using it, and we’re getting a lot of feedback.”
On-time and on-budget release of the new platform.
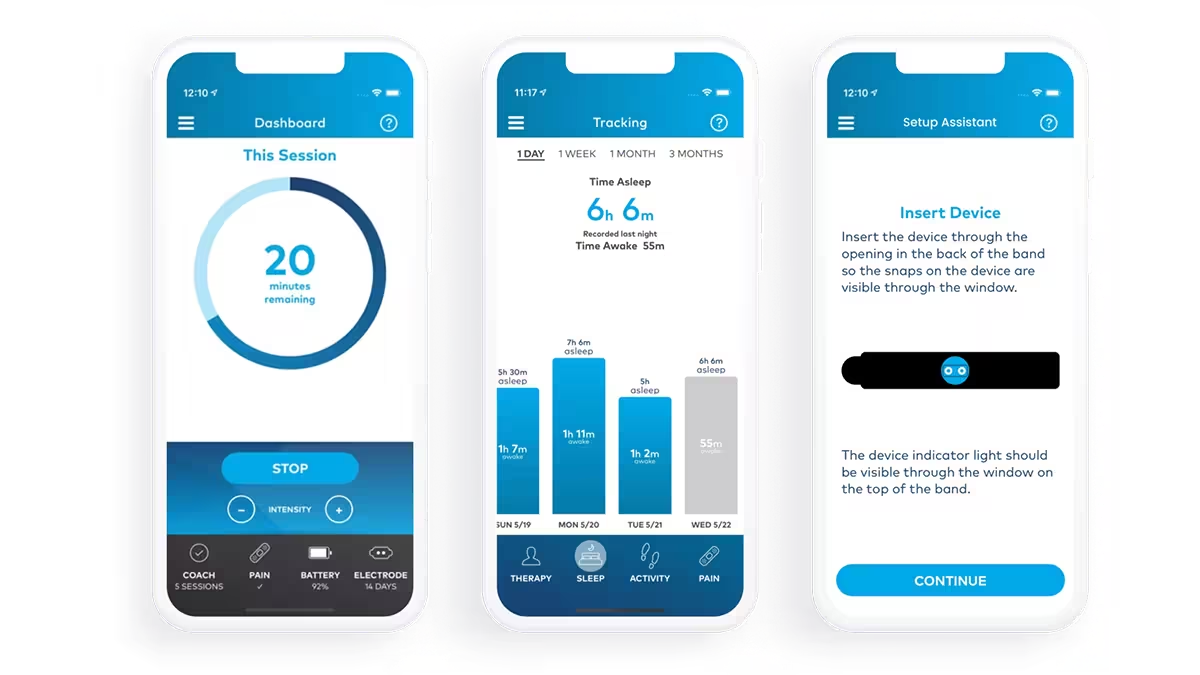
The Quell Relief mobile app lets users customize therapy, track detailed health metrics, and access real-time device status. It syncs seamlessly with the Quell wearable device via Bluetooth LE and supports NeuroMetrix's mission of improving the lives of chronic pain sufferers. Atomic Object delivered a polished, user-friendly app that extends the functionality of Quell, with comprehensive features for therapy management and health insights. IDEO completed the industrial design and mechanical engineering of the Quell medical device.

Making Bluetooth Connectivity User-Friendly
The app's Bluetooth connectivity was designed with users of all abilities in mind. Atomic Object’s project team focused on creating an intuitive connection process that minimized user frustration. Our team performed testing, and designed features like automatic pairing and reconnection, ensuring users could easily sync their Quell device without extensive technical knowledge.
Empowering Users Through Customization
Atomic Object worked closely with NeuroMetrix to deeply understand the device’s sensors and hardware capabilities. By collaborating hands-on with the technology, the group designed and implemented customizable features that allowed users to tailor therapy intensity, track specific health metrics, and schedule sessions. These features helped the Quell Relief app meet diverse user needs while maintaining simplicity in design and functionality.
Supporting Scientific Validation and User Success
Quell's effectiveness was validated in the Journal of Pain Research and highlighted in media outlets like Fast Company and CNET. Studies showed that 81% of users experienced improved health, and 67% reduced reliance on pain medication, solidifying Quell as a groundbreaking solution in pain management. In 2021, the US Food and Drug Administration (FDA) granted Quell a "breakthrough device" designation for the treatment of fibromyalgia symptoms.
Validated, third party reviews from Atomic clients
See how our clients rate their experience working with Atomic on Clutch. Read verified reviews that highlight our commitment to delivering innovative solutions and exceptional results.




